-
News
Our PhD Students, Natalia Hernández and Marcos Sánchez, at the Gene and Cell Teraphy Congress and the Biennial Chemistry Meeting
4 Jul, 2023
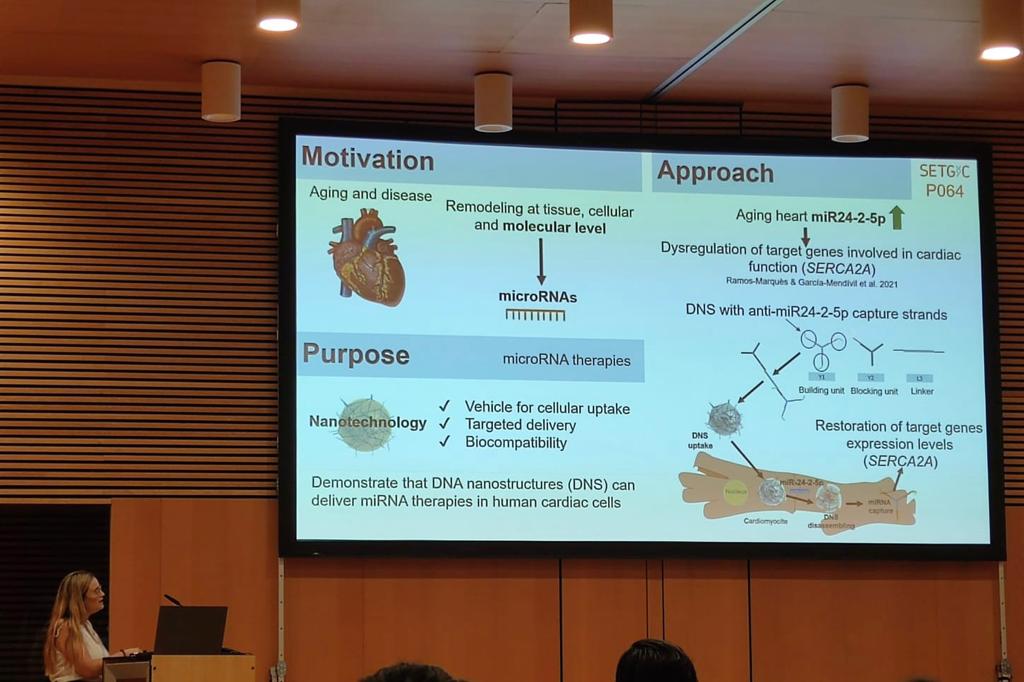
Natalia Hernández Bellido: “Non-viral delivery of microRNA therapies for cardiac dysfunction using DNA nanostructures”
Cardiac microRNAs (miRNAs) are dysregulated in cardiac disease and aging. Thanks to their pleiotropic effects, they are spotlighted as therapeutic targets to treat cardiac conditions. Nanotechnology enables targeted delivery of miRNA therapies, avoiding their low stability and potential off-target effects. Particularly, DNA nanostructures (DNS) for miRNA therapies have not yet been applied to cardiac disease. Our group has identified MIR24-2 upregulated with age in human left ventricle and miR24-2-5p to interact with genes essential in cardiac function, such as SERCA2, which is also downregulated in heart failure.
The main goal of this study is to demonstrate the capacity of DNS to deliver anti-miRNA therapies in human cardiomyocytes.
Different versions of DNS were designed and generated by self-assembly of DNA strands containing anti-miR24-2-5p capture segments. Their physicochemical and structural characterization showed optimal thermal stability at physiological temperature, proper stability in serum and specific disassembly in the presence of miR24-2-5p. Biological characterization of DNS in vitro in Hek293 cells demonstrated high uptake levels with partial degradation after 48h. However, internalization in human induced pluripotent stem cells (iPSC)-derived cardiomyocytes (iCM) was remarkably lower. DNS demonstrated in vitro biocompatibility in both, Hek293 and in iCM.
To conclude, our biocompatible anti-miR-loaded DNS effectively capture miR-24-2-5p in vitro and internalized in iCM, indicating their suitability to carry miRNA therapies to iCM in vitro, although with lower efficiency than in Hek293 cells. Therefore, future efforts will focus on DNS functionalization with ligands that promote their efficient and specific uptake by primary cardiac cells.